The Kx team attended the World Orphan Drug Congress (WODC) in Boston, MA last week and came away energized from three days of sharing ideas, inspirational stories, and panel discussions. This event brings together the full range of stakeholders who drive and support orphan drug development, regulatory approval, treatment delivery, and patient/caregiver support services. Here we discuss a few themes we heard across meetings and presentations:
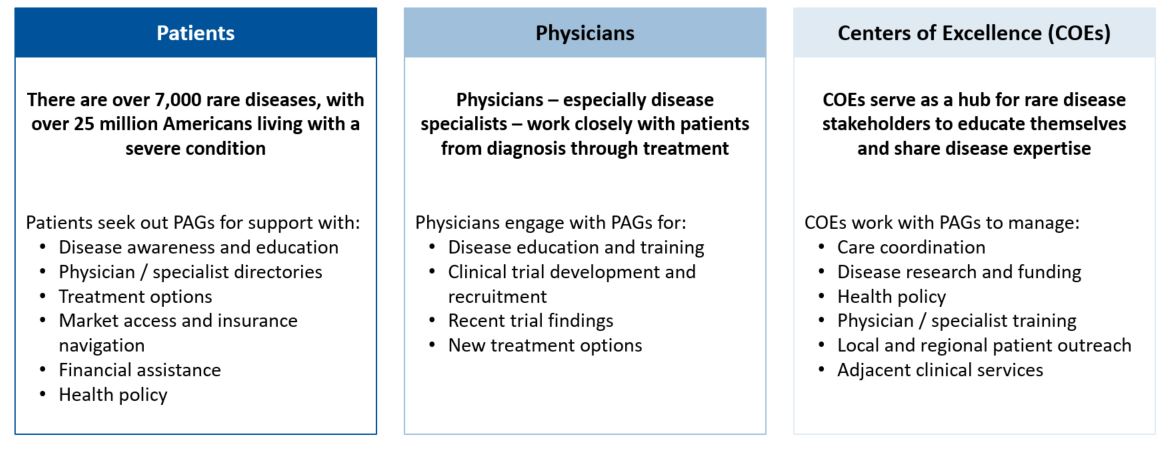
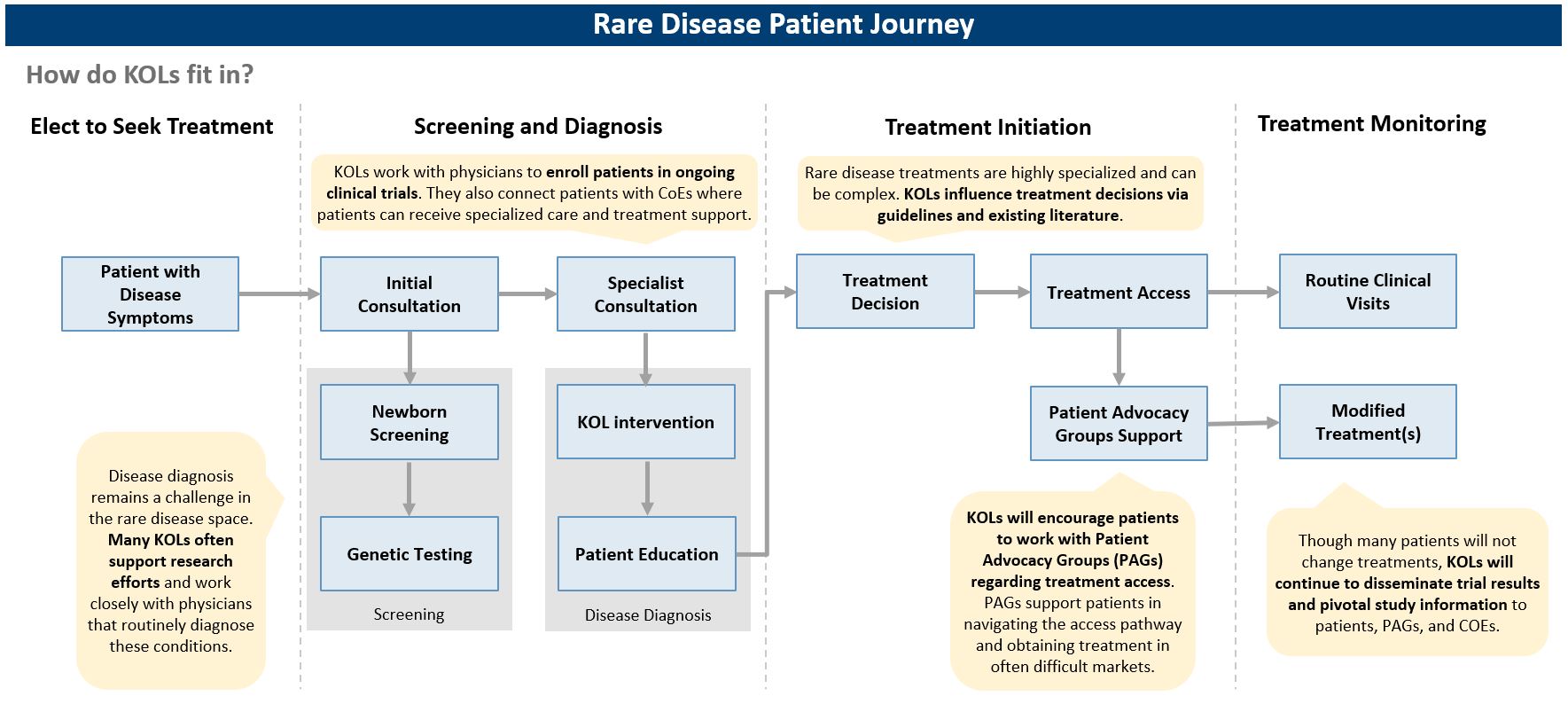
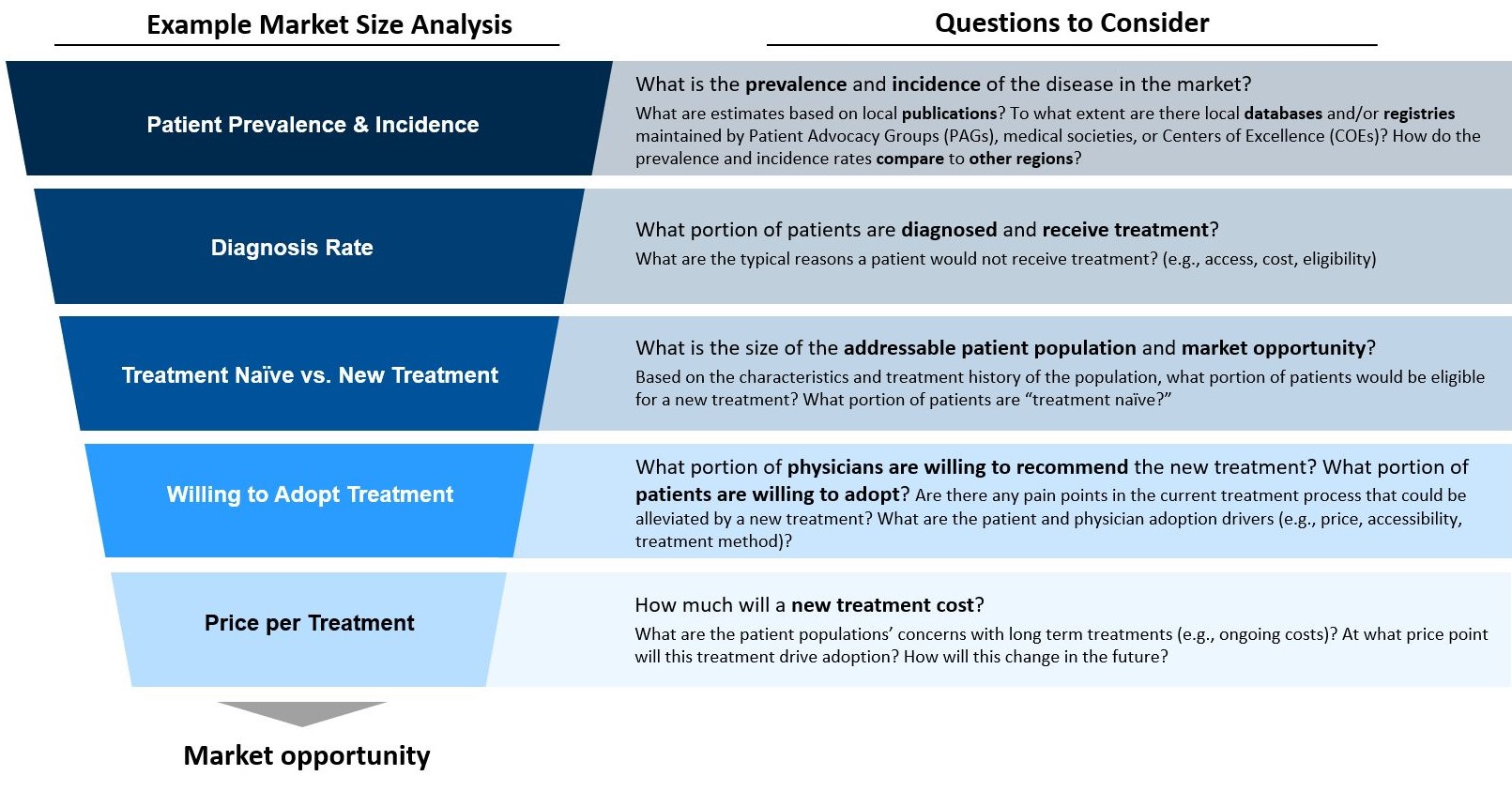
Capturing the full market potential for a new orphan drug is a challenge; diagnosis and treatment gaps add to the unmet needs a new treatment needs to address
Repeatedly, newly approved treatments for rare diseases with a strong clinical profile (i.e., safe and effective) have not achieved the degree of adoption forecasted pre-launch. Why? With humility, drug developers acknowledged their own shortcomings in not building a comprehensive ecosystem of care for target patients, their caregivers, HCPs, and payers.
Unfortunately, there is no universal go-to-market (GTM) playbook for launching a treatment in an orphan indication; however, key questions to ask when GTM planning in rare diseases in have become clear:
- What is the daily reality and emotional state of patients and caregivers?
- What breakpoints exist in the patient journey, and how can we address those?
- How can barriers to delivery be mitigated through just-in-time delivery infrastructure and treatment sites creation for gene therapies?
- How should value be measured (to ensure access and coverage continuation)?
The term “beyond the pill” is still a popular idea, but there is a need to define what that means for meeting the needs of different patient, HCP, and payer populations. By establishing trusting, longitudinal relationships with the patients, caregivers, and HCPs they strive to serve, drug developers are successfully building tailored, comprehensive GTM strategies today for the next wave of orphan drug launches, building the foundation for “beyond the pill” to be more than a catchphrase.
Value differentiation is best achieved through focus on simple, straightforward clinical endpoints
For over a decade, rare disease treatments have seen greater flexibility from regulators on primary endpoint designations than treatments for more prevalent indications; however, we consistently heard that leniency in clinical trial design may fade. Until now, drug developers could drive endpoints selection based on early trial results and carry these through to regulatory approval reviews. As noted by Simone Boselli, Public Affairs Director, EURODIS, during one of the panel discussions, too often today these endpoints are abandoned post-approval given challenges with continued tracking in real-world settings.
Going forward, drug developers should pursue greater prospective consideration and engagement to ascertain how regulatory bodies (FDA, EMA, health technology assessment (HTA) bodies) expect data packages to be constructed. Emphasis will be placed on using clinical endpoints that are simple to compare across studies by competing developers and straightforward to extrapolate to functional benefits for patients, as well as feasible to continue to measure in the real world as drug developers aim to build a real-world evidence base. In the future, drug developers presenting unique (rather than universally accepted) clinical endpoints may run into delays or rejections, costing time, revenue, and patient access to novel treatments.
Rare disease patients desire true partnership, including greater influence over the risk tolerance decisions and transparency on drug development setbacks
Appropriately, keynote presentation sessions often included patient advocates such as Allyson Berent, COO, GeneTx*, who shared her experience from her daughter’s Angelman Syndrome (AS) diagnosis at 6 months old. When she realized there were no approved treatments for the condition, she dedicated her time and energy to pursuing an effective treatment, ultimately becoming the Chief Science Officer of Foundation for Angelman Syndrome and co-founder of GeneTx.
(*note: GeneTx was acquired by Ultragenyx on 7/19/22)
We at Kx Advisors applaud the dedication of individuals, such as Allyson Berent, who work relentlessly to bring innovative therapies to patients. Also, we are telling her story here because her asks to industry were echoed by others at WODC:
- Further elevate patients’ voices and requests – creating an ecosystem of care requires early and frequent patient input
- Consider that the risk-reward tolerance when patients are desperate for a lifeline may be different than industry’s view; many felt patients should have more influence on the calculus
- Increase transparency of critical feedback from the FDA to drug developers when reviewing clinical results, as patients are central to progress and deserve transparency on both advancements and setbacks; this sharing rarely occurs today, and collectively holds back innovation for the sake of competitive advantage
How Kx Can Help
Do you want to discuss further our takeaways from the World Orphan Drug Conference? Does your organization need to address specific challenges in rare disease asset planning and launch strategy? If so, please contact Brett Larson [email protected] or Jenna Riffell at [email protected].